
Effect of ticagrelor with or without aspirin on vein graft outcome 1 year after on-pump and off-pump coronary artery bypass grafting
Introduction
Various factors are suggested to affect the outcomes of coronary artery bypass grafting (CABG), among which, the effects of cardiopulmonary bypass (CPB) has been extensively discussed for years but still controversial (1-3). Studies suggested that the activation and consumption of clotting factors and platelets in CABG with CPB (on-pump CABG) were associated with systemic inflammatory response and poor hemostasis compared with CABG without CPB (off-pump CABG) (1). On the other hand, the clotting disorders and platelet dysfunction with on-pump CABG may increase graft (especially vein graft) patency compared with off-pump surgery (4,5). Therefore, although different guidelines recommend dual antiplatelet therapy (DAPT) after CABG to reduce major adverse cardiovascular events (MACEs) and preserve graft patency in patients with a history of acute coronary syndrome (ACS) (1,6,7), the recommendation levels for off-pump (Class I) and on-pump (Class IIb) are different by considering that the use of CPB during CABG may affect the choices of therapeutic regimen and effects of antiplatelet therapy. In addition, beneficial effects of DAPT on graft patency was suggested not well demonstrated (8). These evidences indicate that the benefits of DAPT are not well established in both on-pump and off-pump CABG.
Moreover, the recommended DAPT regimens after CABG are mostly based on findings on the use of clopidogrel plus aspirin. Ticagrelor, a reversible inhibitor of the P2Y12 receptor, in addition to aspirin, significantly reduced MACE in patients with ACS compared with clopidogrel (9). The CABG substudy from the PLATO trial showed that compared with clopidogrel, ticagrelor was associated with a substantial reduction in all-cause and cardiovascular mortality, without an excess risk of CABG-related bleeding (10). The Different Antiplatelet Therapy Strategy After Coronary Artery Bypass Graft Surgery (DACAB) trial showed that ticagrelor plus aspirin resulted in a significant improvement in vein graft patency 1 year after CABG compared with aspirin alone (11). Compared with the practice in US and Europe, a higher proportion of patients (approximately 76%) in this trial underwent off-pump CABG, revealing the local practice in China, further on in East Asia. Here we examined the effects of ticagrelor with or without aspirin on vein graft patency 1 year after on-pump or off-pump CABG.
We present the following article in accordance with the 2010 CONSORT reporting checklist (available at http://dx.doi.org/10.21037/jtd-20-1177).
Methods
Background
The DACAB trial was a multicenter, randomized, open-label, parallel control study of 500 patients with 1,460 vein grafts undergoing CABG in China. Details about the study design, patients, and results have been published previously (11). The trial was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and the Harmonized Tripartite Guideline for Good Clinical Practice from the International Conference on Harmonization. The trial protocol and all amendments were reviewed and approved by the independent Institutional Review Board responsible for each participating site. Written informed consent was obtained from all individual participants included in the study.
Population
For current post-hoc study, all patients in the DACAB study were included in the analysis. Briefly, patients aged 18–80 years with indications for CABG using vein grafts were eligible for the trial. Major exclusion criteria included urgent revascularization or other concomitant cardiac surgery, a need for dual antiplatelet or vitamin K antagonist therapy after CABG, and serious bleeding risk. Detailed inclusion and exclusion criteria are listed in Table S1.

Full table
Patients were randomly (in blocks of 3) assigned in a 1:1:1 ratio to receive ticagrelor (Brilinta, AstraZeneca) 90 mg twice daily plus aspirin 100 mg (Bayaspirin, Bayer) once daily, T + A regimen; ticagrelor 90 mg twice daily, T regimen; or aspirin 100 mg once daily, A regimen. Both on-pump and off-pump CABG was allowed in the trial and was selected according to the surgeons’ decisions. Only surgeons with the experience of more than 100 off-pump cases annually were invited to participate in the trial. The vein graft was openly harvested. The patients started their allocated regimen within 24 hours after the surgery, and the median duration of study treatment was 1 year.
Outcomes
Consistent with the DACAB trial, the primary outcome of this post hoc analysis was the vein graft patency (stenosis<50%) rate 1 year after CABG. Vein grafts were assessed by multislice computed tomographic angiography or coronary angiography and interpreted by an independent Image Data Review Centre blinded to treatment allocation. Other cardiovascular outcomes includes: (I) the rate of MACE [defined as cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke (12)] 1 year after the surgery; and (II) the rate of relief from angina pectoris evaluated using a questionnaire based on the Canadian Cardiovascular Society grade 1 year after the surgery.
For safety outcomes, the adverse events (AEs) and serious AEs (SAEs) were recorded for 1 year. Bleeding events were classified as those in the DACAB trial. The definitions of these outcomes have been previously reported (11).
Statistical analysis
This post-hoc analysis was to compare vein graft patency 1 year after CABG between patients receiving T + A or T versus A in the on-pump and off-pump patients, respectively. The primary analysis was conducted on a per-graft basis according to the intention-to-treat (ITT) principle. The ITT population included all randomized patients. Missing data on graft outcome were handled as occlusion. One-year patency was also evaluated based on a per-patient basis. A per-patient analysis was performed in which patients were classified according to the graft with the worst degree of stenosis.
Differences in baseline characteristics between treatment groups after on-pump and off-pump CABG, as well as between on-pump and off-pump were compared using one-way analysis of variance or Kruskal-Wallis test for continuous variables and chi-square test or Fisher’s exact test for categorical variables. Generalized estimating equations logistic model and multivariable logistic model were applied to estimate odds ratio (OR) and 95% confidence interval (CI) in per-graft and per-patient analysis, respectively. Outcome was defined as non-patency or occlusion when calculating OR. Confounders adjusted in the multivariable model included age; sex; medical history of hypertension, diabetes, and hyperlipidemia; SYNTAX score; target vessel distribution; and statin use 1 year after CABG. The potential modifying effect of pump status on different antiplatelet strategies was formally tested by including a multiplicative interaction term between pump status and antiplatelet treatment. All statistical analyses were carried out using SAS 9.4 (SAS Institute, NY, USA). Two-sided P values <0.05 were considered statistically significant.
Results
Baseline characteristics
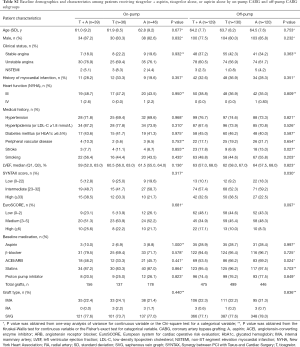
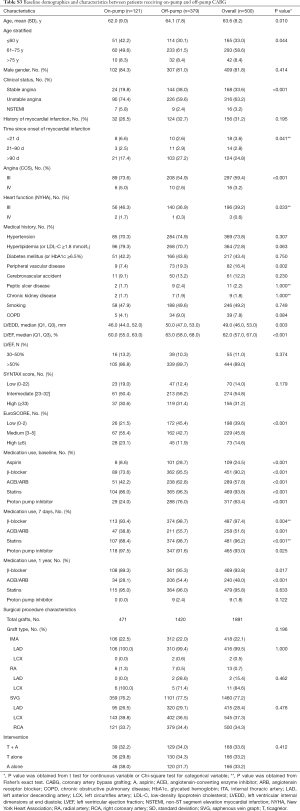
Among 500 patients with 1,460 vein grafts randomized in the trial, 121 patients with 359 vein grafts received on-pump CABG (39 patients with 121 vein grafts in T + A regimen, 36 with 101 vein grafts in T regimen, and 46 with 137 vein grafts in A regimen) and 379 patients with 1,101 vein grafts received off-pump CABG (129 patients with 336 vein grafts in T + A regimen, 130 with 387 vein grafts in T regimen, and 120 with 348 vein grafts in A regimen). The baseline characteristics were generally balanced among the three randomized antiplatelet treatment regimens in either on-pump or off-pump subgroups, except that of the history of stroke, SYNTAX score, and angiotensin-converting enzyme inhibitor (ACEI)/angiotensin receptor blocker (ARB) use among the three antiplatelet treatment regimens in the off-pump subgroup (Table S2). The comparisons on baseline demographics between on-pump and off-pump subgroups are detailed in Table S3.
Full table
Full table
One-year vein graft outcomes
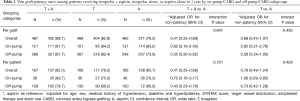
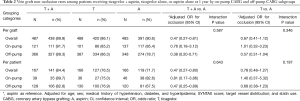
The graft-level analysis showed that the 1-year patency rate of vein graft was 91.7% in T + A regimen compared with 83.2% in A regimen for the on-pump subgroup (adjusted OR for non-patency =0.62; 95% CI, 0.16–2.45), and 87.7% in T + A regimen compared with 73.9% in A regimen for the off-pump subgroup (adjusted OR for non-patency =0.35; 95% CI, 0.20–0.62). As the interaction P between on-pump and off-pump subgroups while comparing T + A versus A was 0.647, the result suggested that T + A was associated with a higher 1-year patency rate of vein grafts compared with A in both on- and off-pump subgroups, which was consistent with the findings of overall trial. On the contrary, no significant differences were observed in the 1-year patency rate of vein grafts between T and A neither in on-pump (adjusted OR for non-patency =0.92; 95% CI, 0.31–2.76) nor in off-pump (adjusted OR for non-patency =0.58; 95% CI, 0.34–1.00) subgroups (P interaction= 0.430). The comparison of 1-year non-occlusion outcome were consistent with the patency outcome, where T + A showed a higher non-occlusion rates compared with A in both off-pump (OR =0.38; 95% CI, 0.20–0.71) and on-pump (OR =0.76; 95% CI, 0.18–3.12) groups with an interaction P=0.587, and no significant differences were found in the comparisons between T and A. Similar results were seen in the patient-level analysis. Detailed vein graft patency and non-occlusion status in graft- and patient-level analyses are summarized in Tables 1 and 2. Comparisons between on-pump and off-pump subgroups are shown in Table S4.
Full table
Full table

Full table
Cardiovascular and bleeding outcomes
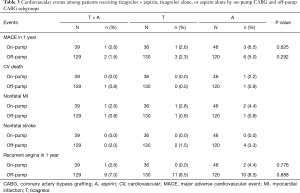
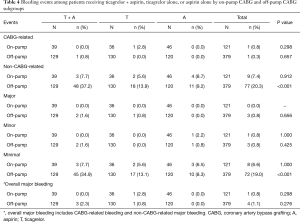
Overall, the incidence of cardiovascular events, including the MACE and recurrent angina within 1 year after the surgery, were relatively low for all three regimens in both on- and off-pump subgroups. In statistical analysis, none of the rates of these events were significantly different among three regimens (Table 3). The bleeding events were similar for the three regimens in the on-pump subgroup; whereas in the off-pump subgroup, the non-CABG-related bleeding rate, particularly minimal bleeding, was significantly higher in T + A compared with A (difference in the rate, 28%; 95% CI, 18.2–37.9) (Table 4). Comparisons between the on- and off-pump subgroups regarding MACE, recurrent angina and bleeding events are shown in Tables S5 and S6.
Full table
Full table

Full table

Full table
Other clinical AEs
Some other clinical AEs, including renal dysfunction (creatinine >200 µmol/L), liver dysfunction (ALT/AST >320/200 U/L), pulmonary infections, and delayed incision healing, are listed in Tables S7 and S8.

Full table

Full table
Discussion
In this post-hoc analysis of the DACAB trial, we aimed to explore the effects of ticagrelor with or without aspirin on vein graft outcomes 1 year after CABG with or without CPB. The results showed a consistent level of benefits of T + A over A alone for both on-pump and off-pump subgroups in increasing vein graft patency. This observation was consistent with the findings of overall DACAB trial, indicating that DAPT with a 12-month T + A regimen may be recommendable for all patients undergoing CABG with vein graft, regardless of the use of CPB.
Life-long aspirin is recognized as the gold standard of care after CABG (13). However, whether additional antiplatelet therapy with a P2Y12 receptor antagonist is beneficial is still controversial (12,14,15). Therefore, recent guidelines suggest that dual antiplatelet treatment should be started after CABG in selected patients (1,6,16). Whether the use of CPB during CABG influences the effects of DAPT needs further investigation. Regarding the effect of on-pump and off-pump CABG on patient outcomes, several studies suggested better graft outcomes in patients undergoing on-pump CABG. Sousa Uva et al. showed a lower graft patency rate for off-pump CABG, but the difference disappeared after controlling the heparin dose (17). Kim et al. demonstrated that the 1-year vein graft patency rate after off-pump CABG was low; they suggested that a specific perioperative anticoagulant therapy could be advisable (18). Gundry et al. reported the re-intervention rates of 30% in the off-pump group and 16% in the on-pump group (19). On the contrary, some other studies showed similar outcomes for the two approaches (2,3). Such contradictory results might be due to the complex prothrombotic response elicited by CABG. Therefore, antithrombosis therapy could be a key to enhancing graft patency, particularly for the hypercoagulated status in off-pump CABG (20,21).
Indeed, a few studies compared the different antiplatelet regimens after on-pump and off-pump CABG. To our knowledge, this post-hoc study is the first clinical report that examined whether the beneficial effect of T + A based DAPT was consistent in patients with or without CPB. The results of this post-hoc study indicated that the T + A regimen was superior in achieving 1-year vein graft patency compared with A alone in both on-pump and off-pump subgroups. On the other hand, clinical outcome was not statistically affected by DAPT, which might due to the insufficient event rates in DACAB trial. As limited analysis, including CURE and PLATO subgroup analysis (22,23), has revealed the benefits of post-CABG DAPT, the potential risks, particularly intracranial bleeding and major gastrointestinal bleeding should be taken into consideration. Therefore, Costa et al. suggested that decision-maker should evaluate the individual patient’s bleeding risk and consider the net clinical benefit from post-revascularization DAPT (24).
Regarding the higher early hypercoagulable state in off-pump than in on-pump CABG, studies suggested that the short-term (1-year) vein graft patency was also affected, resulting in a lower patency rate in off-pump CABG (18,21). However, more randomized trials indicated that the hypercoagulable state did not affect either very short-term (30 days) or short-term (1 year) graft patency; they also showed that on-pump and off-pump CABG achieved similar patency (25). In the very short-term, vein graft failure might be triggered not only by the hypercoagulable state, but more by surgical accuracy, graft and target vessel quality, which could be hardly compensated by sophisticated antiplatelet strategy. In the short term or long term, the effects of antiplatelet strategy may play a greater role. The present study obtained similar results that 1-year patency rates were consistent in on- and off-pump CABG, suggesting a limited effect from the early coagulant state in most patients receiving antiplatelet therapy after CABG. Nevertheless, such conclusions should be confirmed in further trials with patients randomly allocated to on-pump and off-pump groups.
Study limitations
This post-hoc analysis had several limitations. First, as a non-prespecified subgroup analysis, the use of CPB was not randomized, but decided by surgeons instead, making the study inadequately powered to detect a statistically significant difference. Similarly, the insufficiency of statistical power, due to the relatively low event rate, to analyze the risk of bleeding and adverse events should be taken into consideration when interpreting the results that no significant differences were observed on major bleeding and MACEs among different antiplatelet strategy groups. Second, the graft outcome, which was the primary outcome in the present study, might not lead to clinical events. Although studies suggested a relationship between occluded grafts and subsequent outcomes, many graft failures are actually clinically silent (26). Also, the vein graft failure was not found to directly result in subsequent death or myocardial infarction (27).
Conclusions
In the present post-hoc analysis, the benefits of ticagrelor plus aspirin for achieving 1-year vein graft patency are consistent in both on-pump and off-pump subgroups, with particular benefit being seen in the off-pump subgroup. The results suggested that patients with vein graft could benefit from 1-year ticagrelor plus aspirin-based dual antiplatelet therapy regardless of the use of CPB during CABG.
Acknowledgments
We thank Dr. Zheng Ke and MedSci for providing medical writing assistance. The statistical analysis was assisted by Department of Biostatistics, School of Public Health, Fudan University, Shanghai, China. All were compensated for their contributions.
Funding: The study was sponsored by Ruijin Hospital, Shanghai, China, and funded by AstraZeneca.
Footnote
Reporting Checklist: The authors have completed the CONSORT reporting checklist. Available at http://dx.doi.org/10.21037/jtd-20-1177
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jtd-20-1177
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-20-1177). YZ has served as a speaker for AstraZeneca, Johnson & Johnson, Novartis, and Sanofi, also as an investigator on clinical trials sponsored by AstraZeneca, Bayer, Novartis, and Sanofi. HL has served as a speaker for Pfizer. RW has served as a speaker for AstraZeneca and as an investigator on clinical trials sponsored by Bayer. XW has served as a speaker for AstraZeneca and Johnson & Johnson. LH has served as a speaker for Medtronic. QZ has served as a speaker for AstraZeneca, Johnson & Johnson, and Medtronic, and also been an investigator on clinical trials sponsored by AstraZeneca, Bayer, Novartis, and Sanofi. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The trial was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and the Harmonized Tripartite Guideline for Good Clinical Practice from the International Conference on Harmonization. The trial protocol and all amendments were reviewed and approved by the independent Institutional Review Board responsible for each participating site. Written informed consent was obtained from all individual participants included in the study.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kulik A, Ruel M, Jneid H, et al. Secondary prevention after coronary artery bypass graft surgery: a scientific statement from the American Heart Association. Circulation 2015;131:927-64. [Crossref] [PubMed]
- Lamy A, Devereaux PJ, Prabhakaran D, et al. Five-Year Outcomes after Off-Pump or On-Pump Coronary-Artery Bypass Grafting. N Engl J Med 2016;375:2359-68. [Crossref] [PubMed]
- Puskas JD, Williams WH, O'Donnell R, et al. Off-pump and on-pump coronary artery bypass grafting are associated with similar graft patency, myocardial ischemia, and freedom from reintervention: long-term follow-up of a randomized trial. Ann Thorac Surg 2011;91:1836-42; discussion 1842-3. [Crossref] [PubMed]
- Møller CH, Penninga L, Wetterslev J, et al. Off-pump versus on-pump coronary artery bypass grafting for ischaemic heart disease. Cochrane Database Syst Rev 2012.CD007224. [PubMed]
- Khan NE, De Souza A, Mister R, et al. A randomized comparison of off-pump and on-pump multivessel coronary-artery bypass surgery. N Engl J Med 2004;350:21-8. [Crossref] [PubMed]
- Valgimigli M, Bueno H, Byrne RA, et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS. Eur J Cardiothorac Surg 2018;53:34-78. [Crossref] [PubMed]
- Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. EuroIntervention 2019;14:1435-534. [Crossref] [PubMed]
- de Leon N, Jackevicius CA. Use of aspirin and clopidogrel after coronary artery bypass graft surgery. Ann Pharmacother 2012;46:678-87. [Crossref] [PubMed]
- Wallentin L, Becker RC, Budaj A, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2009;361:1045-57. [Crossref] [PubMed]
- Brilakis ES, Held C, Meier B, et al. Effect of ticagrelor on the outcomes of patients with prior coronary artery bypass graft surgery: insights from the PLATelet inhibition and patient outcomes (PLATO) trial. Am Heart J 2013;166:474-80. [Crossref] [PubMed]
- Zhao Q, Zhu Y, Xu Z, et al. Effect of ticagrelor plus aspirin, ticagrelor alone, or aspirin alone on saphenous vein graft patency 1 year after coronary artery bypass grafting: a randomized clinical trial. JAMA 2018;319:1677-86. [Crossref] [PubMed]
- Chakos A, Jbara D, Singh K, et al. Network meta-analysis of antiplatelet therapy following coronary artery bypass grafting (CABG): none versus one versus two antiplatelet agents. Ann Cardiothorac Surg 2018;7:577-85. [Crossref] [PubMed]
- Hillis LD, Smith PK, Anderson JL, et al. 2011 ACCF/AHA Guideline for Coronary Artery Bypass Graft Surgery. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Developed in collaboration with the American Association for Thoracic Surgery, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons. J Am Coll Cardiol 2011;58:e123-210. [Crossref] [PubMed]
- Kulik A, Le May MR, Voisine P, et al. Aspirin plus clopidogrel versus aspirin alone after coronary artery bypass grafting: the clopidogrel after surgery for coronary artery disease (CASCADE) Trial. Circulation 2010;122:2680-7. [Crossref] [PubMed]
- Gao G, Zheng Z, Pi Y, et al. Aspirin plus clopidogrel therapy increases early venous graft patency after coronary artery bypass surgery a single-center, randomized, controlled trial. J Am Coll Cardiol 2010;56:1639-43. [Crossref] [PubMed]
- Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J 2018;39:119-77. [Crossref] [PubMed]
- Sousa Uva M, Cavaco S, Oliveira AG, et al. Early graft patency after off-pump and on-pump coronary bypass surgery: a prospective randomized study. Eur Heart J 2010;31:2492-9. [Crossref] [PubMed]
- Kim KB, Lim C, Lee C, et al. Off-pump coronary artery bypass may decrease the patency of saphenous vein grafts. Ann Thorac Surg 2001;72:S1033-7. [Crossref] [PubMed]
- Gundry SR, Romano MA, Shattuck OH, et al. Seven-year follow-up of coronary artery bypasses performed with and without cardiopulmonary bypass. J Thorac Cardiovasc Surg 1998;115:1273-7; discussion 1277-8. [Crossref] [PubMed]
- Cavalca V, Rocca B, Veglia F, et al. On-pump Cardiac Surgery Enhances Platelet Renewal and Impairs Aspirin Pharmacodynamics: Effects of Improved Dosing Regimens. Clin Pharmacol Ther 2017;102:849-58. [Crossref] [PubMed]
- Bednar F, Osmancik P, Vanek T, et al. Platelet activity and aspirin efficacy after off-pump compared with on-pump coronary artery bypass surgery: results from the prospective randomized trial PRAGUE 11-Coronary Artery Bypass and REactivity of Thrombocytes (CABARET). J Thorac Cardiovasc Surg 2008;136:1054-60. [Crossref] [PubMed]
- Fox KA, Mehta SR, Peters R, et al. Benefits and risks of the combination of clopidogrel and aspirin in patients undergoing surgical revascularization for non-ST-elevation acute coronary syndrome: the Clopidogrel in Unstable angina to prevent Recurrent ischemic Events (CURE) Trial. Circulation 2004;110:1202-8. [Crossref] [PubMed]
- Held C, Asenblad N, Bassand JP, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes undergoing coronary artery bypass surgery: results from the PLATO (Platelet Inhibition and Patient Outcomes) trial. J Am Coll Cardiol 2011;57:672-84. [Crossref] [PubMed]
- Costa F, Van Klaveren D, Feres F, et al. Dual Antiplatelet Therapy Duration Based on Ischemic and Bleeding Risks After Coronary Stenting. J Am Coll Cardiol 2019;73:741-54. [Crossref] [PubMed]
- Puskas JD, Williams WH, Mahoney EM, et al. Off-pump vs conventional coronary artery bypass grafting: early and 1-year graft patency, cost, and quality-of-life outcomes: a randomized trial. JAMA 2004;291:1841-9. [Crossref] [PubMed]
- Harskamp RE, Alexander JH, Schulte PJ, et al. Vein graft preservation solutions, patency, and outcomes after coronary artery bypass graft surgery: follow-up from the PREVENT IV randomized clinical trial. JAMA Surg 2014;149:798-805. [Crossref] [PubMed]
- Lopes RD, Mehta RH, Hafley GE, et al. Relationship between vein graft failure and subsequent clinical outcomes after coronary artery bypass surgery. Circulation 2012;125:749-56. [Crossref] [PubMed]


