At present, robot-assisted lobectomy is one of the most advanced
minimally invasive thoracic surgeries merely conducted in
few medical centers in Europe, USA, Japan, Hongkong and Taiwan
of China. The da Vinci Surgical Robotic System (Intuitive
Surgical, Sunnyvale, California), a FDA (U.S. Food and Drug Administration)
authenticated robotic system, is made up of three
parts: 1. Surgeon console; 2. "Praying mantis-like" robotic arms
chassis, which includes robot optical system and three robotic
arms; 3. Electronic communications tower system connecting main
console and robotic arms (
3). The robotic system our department
applied is the newest da Vinci Surgical System modified from the
former da Vinvi systems. Auxiliary instruments of da Vinci S Surgical Sys tem for thoracoscopic surgery are conveniently attached
on col lapsible robotic arms, which not only reduces robotic arm
external collision, but also extends the robot functional range.
Moreover, da Vinci S Surgical System is an ergonomic operating
system, it provides a high-definition monitor (
4).
Comparing with conventional video-assisted thoracoscopic
surgeries, robot-assisted thoracoscopic surgery owns a three-dimensional
high-definition dynamic visualization, permitting a
clearer and more intuitional vision for surgeons; besides, robotics
retains superiorities such as anti-shake arms, fixed endoscope, wide
range of motion, high mobility of instruments, which make
robot-assisted surgery to be finer. However, one of the significant
shortcomings is the lack of haptic feedback (
5).
Doctors' rich experience in thoracotomy and complete video-assisted
thoracoscopic lobectomy is essential to robot-assisted lobectomy.
Though visualization supplied by robotics is superior, the
surgeon has to "feel" through visual sense due to lack of haptic
feedback, which brings challenge to the surgeon, so that surgeons
are required to be skilled in thoracoscopic anatomy. Besides, since
preoperative debugging and other procedures are rather elaborate,
it's advised to invite correlated technicians from the robot company
to assist the operation in the beginning.
Robotic surgery is different from other surgeries, so prediction
of a longer operative time is required. Either disharmonious positions
between personnel and robot, or improper ports, or inappropriate
patient position might lead to a long and frustrating operation.
For instance, the first robot-assisted lobectomy our department
undertaken, also the first case in China, took 6.5 hours in total.
Patients selected should be in good fitness neither with a thoracic
surgery history nor with evidence indicating a possibility of
pleural adhesion. The tumor should be of peripheral type with
2~3cm in diameter. As the robot-assisted lobectomy cases increase
and operation group are better organized, operation will be performed
quicker.
Since robot-assisted lobectomy is mainly performed by a single
surgeon, theoretically speaking, dependence on the assistant is
weakened. However, assistant is still an essential factor to an operation.
In video-assisted thoracoscopic lobectomy, pulmonary tissue
traction and exposure, the manipulation of automatic stapler are
conducted by assistant under the guidance of surgeon; however,
during a robot-assisted lobectomy, surgeon stands off the operation
table, emergent actions such as establishing additional exposure,
stapler application, fast and accurate hemostasis are taken by assistant
without surgeon's instruction, which requires the assistant experienced
in minimally invasive surgery and being able to conduct
thoracotomy. In addition, other assistants are also essential. Skilled
instrument nurse and circulating nurse can predict what instruments
might be used, and they are experienced in installing and replacing
instruments, which can save operative time. In case of accidents,
instruments for routine thoracotomy are prepared during operation,
and so it is with gauze attached on forceps. A well-organized group benefits an operation to be smoothly undertaken.
Lymph node dissection is an essential factor of surgery for lung
cancer. Owning to robotics' better visualization and fine maneuver,
mediastinal, aortic-pulmonary arterial, hilar, subcarinal and paratracheal
nodal dissection by robotics is safer and more thorough.
Though existence of aortic arch brings difficulty to left paratracheal
and parabronchial nodal dissection, flexibility of robotics
makes it possible to be completed.
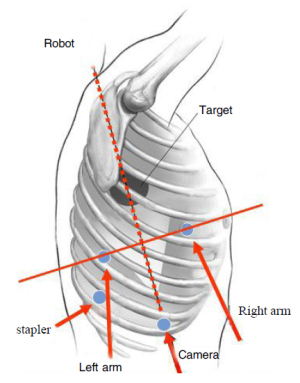
According to literature, there are 3~4 incisions in most complete
video-assisted thoracoscopic surgery lobectomies, among
these incisions, one is for endoscope, the other 2~3 are primary and
auxiliary ports. However, since ends of robotic surgical instruments
are rather fine, relatively "extensive" common thoracoscopic pulmonary
forceps are applied for traction of pulmonary tissues.
Moreover, aspirator for aspirating smoke and blood is important as
well. So far, stapling of vessels, bronchi and so on still relies on automatic
stapler but not corresponding robotic instruments. According
to literature, additional 1~3 auxiliary ports are required for assisting
the operation besides the observation port and ports for
robotic left and right arms. Our initial experience is to establish an
auxiliary port behind observation port for insertion of pulmonary
forceps, automatic stapler and aspirator, etc. Meanwhile, either aspirator
or hem-o-lok clip can be introduced together with robotic
right arm through the bigger incision. Left and right arm ports
should be more cautiously and exactly positioned than video-assisted
thoracoscopic surgery because the distance between the two
ports is required to be long enough to avoid robotic arms collision.
In conclusion, our experience has verified that lobectomy and systemic
lymph node dissection can be accomplished through
robot-assisted thoracoscopic surgery, which treats early stage
non-small cell lung cancer normatively and releases patient's suffering
maximally. Though robotics is developing, lots of its techniques
are still not well developed. As time passes by and technology
develops, robotic performance would be further improved after
full acknowledgement of robotic performance and proficient cooperation
between operator and robots. Robot-assisted minimally invasive
thoracic surgery is leading the way of thoracic surgery.