Prognostic value of COX-2 expression in patients with non-small cell lung cancer: a systematic review and meta-analysis
Introduction
Lung cancer remains the most lethal cancer worldwide, despite improvements in diagnostic and therapeutic techniques. Its incidence has not peaked in many parts of world, particularly in China, which has become a major public health challenge all the world (1). The prognosis for lung cancer patients is generally poor, with an overall 5-year survival rate of approximately 15%, and it has shown little improvement in recent decades (2,3). Several independent prognostic factors for survival have been identified: performance status (PS), disease stage, age, sex and amount of weight lost (4). Some of these factors are useful when choosing treatment options for an individual, principally disease stage and PS. However, the discriminant value of most potential prognostic biological markers is insufficient to predict the optimal therapeutic course for an individual (5,6).
Epidemiologic studies and meta-analysis have shown that prolonged use of non-steroidal anti-inflammatory drugs (NSAIDs) reduces the risk of several solid tumor including bladder cancer, esophageal carcinoma and lung cancer (7-9), and recent meta-analysis suggests that low-dose aspirin could reduce the relative risk of cancer mortality (10). The best-known target of NSAIDs, including aspirin, is the enzyme cyclooxygenase (COX), a key enzyme involved in the production of prostaglandins and other eicosanoids from arachidonic acid. Two COX isoforms, COX-1 and COX-2,
have been identified. Whereas COX-1 is considered a constitutively expressed housekeeping gene, COX-2 is an inducible immediate-early gene associated with inflammation and carcinogenesis (11-13).
The association between COX-2 overexpression and survival in lung cancer patients has been studied for over a decade. However, no consensus has been reached; conflicting results have been reported from different laboratories. We therefore carried out a meta-analysis of data from published studies to quantitatively review the effect of COX-2 overexpression in tumor tissue on survival in patients with non-small cell lung cancer (NSCLC).
Materials and methods
Search strategy and study selection
The electronic databases PubMed, Embase, and CNKI (China National Knowledge Infrastructure) were searched for studies to include in the present meta-analysis. An upper date limit of Jan 1, 2013 was applied; we used no lower date limit. Searches included the terms “lung cancer”, “cyclooxygenase”, “cyclooxygenase-2”, “COX-2” and “prognosis”. We also reviewed the Cochrane Library for relevant articles. The references reported in the identified studies were also used to complete the search.
Studies eligible for inclusion in this meta-analysis met the following criteria: (I) measure COX-2 expression in the primary lung cancer tissue with IHC (immunohistochemistry) or RT-PCR (reverse transcription-polymerase chain reaction); (II) provide information on survival (studies investigating response rates only were excluded); (III) have a follow up time exceeding 5 years; and (IV) When the same author reported results obtained from the same patient population in more than one publication, only the most recent report, or the most complete one, was included in the analysis. Two reviewers (PZ and QQ) independently determined study eligibility. Disagreements were resolved by consensus.
Data extraction and quality assessment
The final articles included were assessed independently by two reviewers (PZ and QQ). Data retrieved from the reports included author-publication year, patient source, histology, disease stage, number of patients, test method, cut-off value, COX-2 positive and survival data (Table 1). If data from any of the above categories were not reported in the primary study, items were treated as “not applicable”. We did not contact the author of the primary study to request the information.
Table1
| First author-year | Patients source | Histology | Stage | N pts | Method | Definition of positivity | Positive (%) | HR estimation | Survival results |
|---|---|---|---|---|---|---|---|---|---|
| Kim SJ-2010 | USA | NSCLC | I-II | 77 | IHC | 5% | 45/77 58% | Surv. curves 0.84 (0.37-1.93) |
NS |
| Tsubochi H-2006 | Japan | NSCLC | I-IIIB | 219 | IHC | Intensity > ref | 137/219 63% | HR 1.60 (0.82-3.15) |
NS |
| Yuan A-2005 | China | NSCLC | I-IV | 60 | RT-PCR | Ratio ref. | 30/60 50% | Surv. curves 6.26 (2.26-9.84) |
poor |
| Laga AC-2005 | USA | NSCLC | I-IV | 259 | IHC | Score 3 | 136/259 52.5% | Surv. curves 0.72 (0.5-1.03) |
NS |
| Richardson CM-2005 | UK | NSCLC | I-IIIA | 172 | IHC | >50% | 91/172 52.9% | Surv. curves 1.12 (0.79-1.60) |
NS |
| Lu C-2004 | USA | NSCLC | I | 94 | ISH | 1% | 56/94 60% | HR 1.80 (1.09-2.96) |
poor |
| Yamaguchi NH-2004 | Brazil | AC | I-IIIB | 117 | IHC | Score 5 | 71/117 60.7% | Surv. curves 0.53 (0.21-0.84) |
Favorable |
| Araki K-2004 | Japan | AC | I | 71 | IHC | >10% | 57/71 80.3% | Surv. curves 1.49 (1.12-6.20) |
poor |
| Kim HS-2003 | South Korea | NSCLC | I-IIIA | 84 | IHC | Score 2 | 67/84 80.0% | Surv. curves 1.88 (1.23-3.26) |
poor |
| Brabender J-2002 | Germany | NSCLC | I-IV | 89 | RT-PCR | Ratio ref. | 47/89 52.8% | Surv. curves 2.26 (1.24-3.67) |
poor |
| Khuri FR-2001 | USA | NSCLC | I | 160 | ISH | 1% | 96/160 60% | Surv. Curves 1.30 (0.93-1.82) |
NS |
| Achiwa H-1999 | Japan | AC | I-IIIB | 130 | IHC | Intensity > ref | 93/130 71.5% | Surv. curves 2.50 (0.95-6.61) I 1.42 (0.88-2.79) ALL |
NS |
| Tian F-2003 | China | NSCLC | I-IV | 79 | IHC | 10% | 54/79 68% | Surv. curves 1.86 (0.8-4.32) |
NS |
| Sun LM-2007 | China | NSCLC | I-III | 116 | IHC | 5% | 78/116 67.2% | Surv. curves 4.95 (2.57-9.52) |
poor |
| Hu XJ-2006 | China | NSCLC | I-III | 88 | IHC | 10% | 63/88 71.6% | Surv. curves 0.53 (0.25-1.14) |
NS |
| Zhang HZ-2005 | China | NSCLC | I-IV | 77 | IHC | 10% | 29/77 37.7% | Surv. curves 0.88 (0.54-1.45) |
NS |
COX-2, Cyclooxygenase-2; IHC, immunohistochemistry; RT-PCR, reverse transcription-polymerase chain reaction; AC, adenocarcinoma; NS, not significant; NA, not applicable; HR, hazard ratio; ISH, in situ hybridisation; N pts, number of patients; NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer; ref, reference; surv. curves, survival curves; NS, non significative; score 2, 3, 4, 5, different scores with combination of percentage of positives cells and intensity.
Statistical methods
For the quantitative aggregation of the survival results, hazard ratios (HR) and their 95% confidence intervals (CIs) were combined to give the effective value. When these statistical variables were not given explicitly in an article, they were calculated from available numerical data using methods reported by Parmar et al. (14).
Heterogeneity of the individual HRs was calculated with Chi-squared tests according to Peto’s method (15). Meanwhile, Heterogeneity test with I2 statistic and Q statistic was performed. All the studies included were categorized by histology, disease stage, patient race. Individual meta-analysis was conducted in each subgroup. If HRs were found to have fine homogeneity, a fixed effect model was used for secondary analysis; if not, a random-effect model was used. In this meta-analysis, DerSimonian-Laird random effects analysis (16) was used to estimate the effect of COX-2 overexpression on survival. By convention, an observed HR>1 implies worse survival for the group with COX-2 overexpression. The impact of COX-2 on survival was considered to be statistically significant if the 95% confidence interval (CI) did not overlap with 1. Horizontal lines represent 95% CIs. Each box represents the HR point estimate, and its area is proportional to the weight of the study. The diamond (and broken line) represents the overall summary estimate, with CI represented by its width. The unbroken vertical line is set at the null value (HR=1.0).
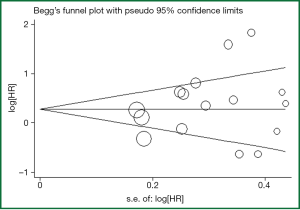
Evidence of publication bias was sought using the methods of Egger et al. (17) and of Begg et al. (18). Moreover, funnel plot (19) was performed to aid in interpreting the funnel plot. If studies appear to be missing in areas of low statistical significance, then it is possible that the asymmetry is due to publication bias. If studies appear to be missing in areas of high statistical significance, then publication bias is a less likely cause of the funnel asymmetry. Intercept significance was determined by the t-test suggested by Egger (P<0.05 was considered representative of statistically significant publication bias). All calculations were performed using STATA version 11.0 (Stata Corporation, College Station, TX).
Results
Study selection and characteristics
Sixteen studies (20-35) published were eligible for this meta-analysis. All reported the prognostic value of COX-2 status for survival in NSCLC patients. The total number of patients included was 1,892, ranging from 60 to 259 patients per study (median 75). The major characteristics of the 16 eligible publications are reported in Table 1.
The included studies considered either all NSCLC subtypes (n=13) and adenocarcinomas (n=3). Four studied reported the information for the stage I disease of all studies. Twelve studies used immunohistochemistry (IHC) to evaluate COX-2 expression in NSCLC, 2 studies used reverse transcription-polymerase chain reaction (RT-PCR) to assess mRNA overexpression in NSCLC, and 2 studies used in situ hybridization (ISH) to determine COX-2 expression. Among the 16 studies, 9 studies were performed in Asian populations, and the remaining 7 studies followed European or American patients. Six of the 16 studies identified COX-2 overexpression as an indicator of poor prognosis, and the other 9 studies showed no statistically significant impact of COX-2 overexpression on survival and only one for favorable prognosis. The proportion of patients exhibiting COX-2 overexpression in individual studies ranged from 37.7% to 80.3% by IHC, from 50% to 52.8% by RT-PCR, and 60% by ISH.
Meta-analysis
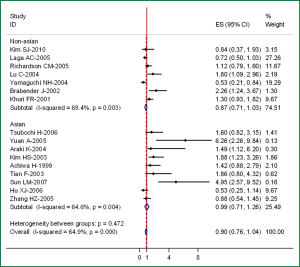
The results of the meta-analysis are reported in Table 2 and in Figure 1. Overall, the combined HR for all 16 eligible studies evaluated COX-2 expression in NSCLC was 0.90 (95% CI: 0.76-1.04), indicating that COX-2 overexpression was not associated with a significant impact on survival. However, highly significant heterogeneity was detected among these studies (I2=64.9%, P=0.000). When grouped according to the geographic settings of individual studies, the combined HRs of Asian studies and non-Asian studies were 0.99 (95% CI: 0.71-1.26) and 0.87 (95% CI: 0.71-1.03), respectively (Figure 1). When grouped according to the method of COX-2 detection used, the combined HR was 0.83 (0.72-1.08) for IHC and 3.28 (1.48-5.13) for RT-PCR, suggesting that COX-2 overexpression is associated with low survival rates for mRNA expression.
Table 2
| Nb Patients | Random effects HR (95% CI) | χ2 heterogeneity test (P) | |
|---|---|---|---|
| Overall | 16 | 0.90 (0.76-1.04) | 0.000 |
| Asian | 9 | 0.99 (0.71-1.26) | 0.004 |
| Non-Asian | 7 | 0.87 (0.71-1.03) | 0.003 |
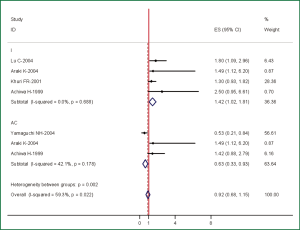
| Adenocarcinoma | 3 | 0.63 (0.33-0.93) | 0.178 |
| Stage I | 4 | 1.42 (1.02-1.81) | 0.688 |
| IHC | 12 | 0.83 (0.72-1.08) | 0.003 |
| PT-PCR | 2 | 3.28 (1.48-5.13) | 0.135 |
Abbreviations: HR, hazard ratio; Nb, number of studies.
The data extracted were adequate to aggregate the studies of stage I NSCLC and lung adenocarcinoma for subgroup analyses. We found one significant correlation, between COX-2 expression and stage I NSCLC. When we aggregated 4 studies that reported results for stage I NSCLC, the combined HR was statistically significant: HR 1.42 (95% CI: 1.02-1.81, P=0.688 for heterogeneity) (Figure 2). We also observed a statistically significant effect of COX-2 expression on survival in lung adenocarcinoma patients with an HR of 0.63 (95% CI: 0.33-0.93, P=0.178 for heterogeneity) (Figure 2), indicating that COX-2 overexpression was a favorable impact on survival.
Publication bias
Begg’s funnel plot and Egger’s test were performed to assess the publication bias in the literature. All 16 eligible studies investigating NSCLC patients yielded a Begg’s test score of P=0.62 and an Egger’s test score of P=0.309, meanwhile according to the funnel plot (Figure 3), the absence of publication bias was found in all 16 studies. These results suggest that there is no publication bias at work.
Discussion
The search for a potential prognostic role of COX-2 in survival for patients with lung cancer is based on its frequent over-expression in NSCLC and also on its potential interference with most pathways implicated in lung carcinogenesis. The role of COX-2 in oncogenesis has widely been studied by in vitro experiments and by in vivo analyses based on animal models. In lung cancer, COX-2 overexpression is associated with micro-vascular angiogenesis (36) and resistance to apoptosis (37). Cyclooxygenase-2 overexpression also decreases host immunity (38) and alters cell adhesion with enhancement of invasion and metastasis (39).
In the present systematic review and meta-analysis, we have combined 16 published studies including 1,892 patients with NSCLC to yield summary statistics indicate that COX-2 overexpression was not associated with a significant impact on survival. When the analysis was restricted to stage I disease, we observed a statistically significant detrimental effect of COX-2 on survival, suggesting that this prognostic factor could be of importance in early-stage NSCLC. In subgroup analysis according to the different techniques used to detect COX-2, results were only significant with RT-PCR.
Despite all these experimental observations, our meta-analysis failed to demonstrate in univariate analysis a statistically significant impact of COX-2 expression as a prognostic factor for overall survival in patients with NSCLC. In subgroup analysis, we observed a significant effect in stage I disease. For early lung cancer overexpressing COX-2 would be more aggressive and would have a worse prognosis than those without COX-2 abnormality. These data could be helpful to determine among stage I diseases those who could benefit from a more aggressive treatment. But the present results concerning the prognostic role of COX-2 in stage I NSCLC still need to be confirmed by adequately designed prospective studies with multivariate analysis before a potential clinical application.
Recently, several systematic reviews (40-48) with meta-analyses on other biological prognostic factors for NSCLC had been reported. P53, microvessel density, HER-2, Ki-67 and RAS might be poor prognostic factors for survival in NSCLC, however, Bcl-2 might be better prognostic factor for survival in NSCLC. In order to clarify the prognostic impact of other biological factors in lung cancer, our group has performed several systematic reviews of the literature with meta-analyses. We found that VEGF (49), E-cadherin (50) and matrix metalloproteinase 2 (51) might be poor prognostic factor in NSCLC, the ground glass opacity (GGO) area (52) had a favorable prognostic value of overall survival and relapse-free survival in small lung adenocarcinoma.
Our data were consistent with the results of a previous meta-analysis (53) published in 2006 that showed a slight detrimental effect on survival in patients with lung cancer is associated with COX-2 expression, but the statistical significance is not reached. That analysis (53) included only 10 studies, and the data were insufficient to determine the prognostic value of COX-2 for subgroups divided according to histology, disease stage and method of COX-2 detection. We have improved upon that previous meta-analysis by including more recent related studies and by generally using a more comprehensive search strategy, screening and study selection were performed independently and reproducibly by two reviewers. We also explored heterogeneity and potential publication bias in accordance with published guidelines.
This systematic review with meta-analysis was complicated by heterogeneity issues. We found highly significant heterogeneity among all studies included. When the analysis was limited to the 3 studies including only adenocarcinomas or 4 studies including only stage I NSCLC, the heterogeneity was not detected. Therefore, histological type and disease stage were not a major source of heterogeneity. The heterogeneity in this study could be explained by the patient source or by differences in the method used to detect COX-2 status. Twelve of the studies included in our analysis used IHC to detect COX-2, and 2 used RT-PCR. When analyzed separately, heterogeneity was still found in the 12 studies that used IHC; however, no heterogeneity was found among the 2 studies that used RT-PCR.
Another potential source of bias is related to the method of HR and 95% CI extrapolation. If these statistics were not reported by the authors, we calculated them from the data available in the article. If this was not possible, we extrapolated them from the survival curves, necessarily making assumptions about the censoring process. Data for multivariate survival analysis reported in the article were included in the present systematic review with meta-analysis; if these data were not available, data calculated from survival curves by univariate analysis were included. These results should be confirmed by an adequately designed prospective study. Furthermore, the exact value of COX-2 overexpression status needs to be determined by appropriate multivariate analysis. Unfortunately, few prospectively designed prognostic studies concerning biomarkers have been reported; thus, our collection of many retrospective studies revealed more significance.
Publication bias (54) is a major concern for all forms of meta-analysis; positive results tend to be accepted by journals, while negative results are often rejected or not even submitted. The present analysis does not support publication bias; the obtained summary statistics likely approximate the actual average. However, it should be noted that our meta-analysis could not completely exclude biases. For example, the study was restricted to papers published in English and Chinese, which probably introduced bias.
In conclusion, there is not prognostic association between COX-2 overexpression and overall survival in patients with NSCLC, but there is a high heterogeneity between the studies. Interestingly, our meta-analysis showed that COX-2 has a detrimental effect on survival in stage I NSCLC. This prognostic role of COX-2 at earliest stage of NSCLC could be of clinical interest in the selection of the patients eligible for induction or adjuvant chemotherapy. Hazard ratio was also significant for the studies using RT-PCR and not for those using IHC, suggesting that a better standardisation of the technique to define and to detect COX-2 positivity is required to the generalisability of the results. Our results should be confirmed by an adequately designed prospective study.
Acknowledgements
This work was supported in part by a grant from “Twelve-Five Plan” the Major Program of Nanjing Medical Science and Technique Development Foundation (Molecular Mechanism Study on Metastasis and Clinical Efficacy Prediction of Non-small Cell Lung Cancer) (Lk-Yu) and Third Level Training Program of Young Talent Project of Nanjing Health (P-Zhan).
Disclosure: The authors declare no conflict of interest.
References
- Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin 2010;60:277-300. [PubMed]
- Molina JR, Yang P, Cassivi SD, et al. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc 2008;83:584-94. [PubMed]
- Alberg AJ, Ford JG, Samet JM, Epidemiology of lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition). Chest 2007;132:29S-55S.
- Paesmans M, Sculier JP, Libert P, et al. Prognostic factors for survival in advanced non-small-cell lung cancer: univariate and multivariate analyses including recursive partitioning and amalgamation algorithms in 1,052 patients. The European Lung Cancer Working Party. J Clin Oncol 1995;13:1221-30. [PubMed]
- Donnem T, Bremnes RM, Busund LT, et al. Gene expression assays as prognostic and predictive markers in early stage non-small cell lung cancer. J Thorac Dis 2012;4:212-3. [PubMed]
- Osarogiagbon RU. Predicting survival of patients with resectable non-small cell lung cancer: Beyond TNM. J Thorac Dis 2012;4:214-6. [PubMed]
- Daugherty SE, Pfeiffer RM, Sigurdson AJ, et al. Nonsteroidal antiinflammatory drugs and bladder cancer: a pooled analysis. Am J Epidemiol 2011;173:721-30. [PubMed]
- Sun L, Yu S. Meta-analysis: non-steroidal anti-inflammatory drug use and the risk of esophageal squamous cell carcinoma. Dis Esophagus 2011;24:544-9. [PubMed]
- Xu J, Yin Z, Gao W, et al. Meta-analysis on the association between nonsteroidal anti-inflammatory drug use and lung cancer risk. Clin Lung Cancer 2012;13:44-51. [PubMed]
- Mills EJ, Wu P, Alberton M, et al. Low-dose aspirin and cancer mortality: a meta-analysis of randomized trials. Am J Med 2012;125:560-7. [PubMed]
- Vane JR, Bakhle YS, Botting RM. Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol 1998;38:97-120. [PubMed]
- Eberhart CE, Coffey RJ, Radhika A, et al. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994;107:1183-8. [PubMed]
- Oshima M, Dinchuk JE, Kargman SL, et al. Suppression of intestinal polyposis in Apc delta716 knockout mice by inhibition of cyclooxygenase 2 (COX-2). Cell 1996;87:803-9. [PubMed]
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med 1998;17:2815-34. [PubMed]
- Yusuf S, Peto R, Lewis J, et al. Beta blockade during and after myocardial infarction: an overview of the randomized trials. Prog Cardiovasc Dis 1985;27:335-71. [PubMed]
- DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177-88. [PubMed]
- Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34. [PubMed]
- Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994;50:1088-101. [PubMed]
- Palmer TM, Peters JL, Sutton AJ, et al. Contour-enhanced funnel plots for meta-analysis. The Stata Journal 2008;8:242-54.
- Kim SJ, Rabbani ZN, Dong F, et al. Phosphorylated epidermal growth factor receptor and cyclooxygenase-2 expression in localized non-small cell lung cancer. Med Oncol 2010;27:91-7. [PubMed]
- Tsubochi H, Sato N, Hiyama M, et al. Combined analysis of cyclooxygenase-2 expression with p53 and Ki-67 in nonsmall cell lung cancer. Ann Thorac Surg 2006;82:1198-204. [PubMed]
- Yuan A, Yu CJ, Shun CT, et al. Total cyclooxygenase-2 mRNA levels correlate with vascular endothelial growth factor mRNA levels, tumor angiogenesis and prognosis in non-small cell lung cancer patients. Int J Cancer 2005;115:545-55. [PubMed]
- Laga AC, Zander DS, Cagle PT. Prognostic significance of cyclooxygenase 2 expression in 259 cases of non-small cell lung cancer. Arch Pathol Lab Med 2005;129:1113-7. [PubMed]
- Richardson CM, Richardson D, Swinson DE, et al. Cyclooxygenase-2 protein levels are independent of epidermal growth factor receptor expression or activation in operable non-small cell lung cancer. Lung Cancer 2005;48:47-57. [PubMed]
- Lu C, Soria JC, Tang X, et al. Prognostic factors in resected stage I non-small-cell lung cancer: a multivariate analysis of six molecular markers. J Clin Oncol 2004;22:4575-83. [PubMed]
- Yamaguchi NH, Lichtenfels AJ, Demarchi LM, et al. COX-2, MMP-9, and Noguchi classification provide additional prognostic information about adenocarcinoma of the lung. A study of 117 patients from Brazil. Am J Clin Pathol 2004;121:78-86. [PubMed]
- Araki K, Hashimoto K, Ardyanto TD, et al. Co-expression of Cox-2 and EGFR in stage I human bronchial adenocarcinomas. Lung Cancer 2004;45:161-9. [PubMed]
- Kim HS, Youm HR, Lee JS, et al. Correlation between cyclooxygenase-2 and tumor angiogenesis in non-small cell lung cancer. Lung Cancer 2003;42:163-70. [PubMed]
- Brabender J, Park J, Metzger R, et al. Prognostic significance of cyclooxygenase 2 mRNA expression in non-small cell lung cancer. Ann Surg 2002;235:440-3. [PubMed]
- Khuri FR, Wu H, Lee JJ, et al. Cyclooxygenase-2 overexpression is a marker of poor prognosis in stage I non-small cell lung cancer. Clin Cancer Res 2001;7:861-7. [PubMed]
- Achiwa H, Yatabe Y, Hida T, et al. Prognostic significance of elevated cyclooxygenase 2 expression in primary, resected lung adenocarcinomas. Clin Cancer Res 1999;5:1001-5. [PubMed]
- Tian F, Wang TY, Gong M, et al. Overexpression of COX-2 and its clinical significance in non-small cell lung cancer. Zhonghua Wai Ke Za Zhi 2003;41:407-10. [PubMed]
- Sun L, Wang L, Fang J, et al. Prognostic significance of the expression of Cox-2 in lung cancer Chin J Cancer Prev Treat 2007;24:1789-91. (in Chinese).
- Hu X, Song N, Liu Y, et al. Prognostic value of Bcl-2 and Cox-2 expressions in non-small cell lung cancer: a tissue microarray analysis of 88 cases. Chin J Cancer Prev Treat 2006;13:1704-8.
- Zhang HZ, Hua P, Li HG, et al. Expression of vascular endothelial growth factor C and cyclooxygenase-2 in non-small-cell lung carcinoma and their clinical significance. Zhonghua Zhong Liu Za Zhi 2005;27:734-7. [PubMed]
- Masferrer JL, Leahy KM, Koki AT, et al. Antiangiogenic and antitumor activities of cyclooxygenase-2 inhibitors. Cancer Res 2000;60:1306-11. [PubMed]
- Hida T, Kozaki K, Muramatsu H, et al. Cyclooxygenase-2 inhibitor induces apoptosis and enhances cytotoxicity of various anticancer agents in non-small cell lung cancer cell lines. Clin Cancer Res 2000;6:2006-11. [PubMed]
- Huang M, Stolina M, Sharma S, et al. Non-small cell lung cancer cyclooxygenase-2-dependent regulation of cytokine balance in lymphocytes and macrophages: up-regulation of interleukin 10 and down-regulation of interleukin 12 production. Cancer Res 1998;58:1208-16. [PubMed]
- Tsujii M, Kawano S, DuBois RN. Cyclooxygenase-2 expression in human colon cancer cells increases metastatic potential. Proc Natl Acad Sci U S A 1997;94:3336-40. [PubMed]
- Steels E, Paesmans M, Berghmans T, et al. Role of p53 as a prognostic factor for survival in lung cancer: a systematic review of the literature with a meta-analysis. Eur Respir J 2001;18:705-19. [PubMed]
- Meert AP, Paesmans M, Martin B, et al. The role of microvessel density on the survival of patients with lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer 2002;87:694-701. [PubMed]
- Meert AP, Martin B, Paesmans M, et al. The role of HER-2/neu expression on the survival of patients with lung cancer: a systematic review of the literature. Br J Cancer 2003;89:959-65. [PubMed]
- Martin B, Paesmans M, Berghmans T, et al. Role of Bcl-2 as a prognostic factor for survival in lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer 2003;89:55-64. [PubMed]
- Martin B, Paesmans M, Mascaux C, et al. Ki-67 expression and patients survival in lung cancer: systematic review of the literature with meta-analysis. Br J Cancer 2004;91:2018-25. [PubMed]
- Mascaux C, Iannino N, Martin B, et al. The role of RAS oncogene in survival of patients with lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer 2005;92:131-9. [PubMed]
- Meert AP, Martin B, Delmotte P, et al. The role of EGF-R expression on patient survival in lung cancer: a systematic review with meta-analysis. Eur Respir J 2002;20:975-81. [PubMed]
- Nakamura H, Kawasaki N, Taguchi M, et al. Survival impact of epidermal growth factor receptor overexpression in patients with non-small cell lung cancer: a meta-analysis. Thorax 2006;61:140-5. [PubMed]
- Fan J, Wang L, Jiang GN, et al. The role of survivin on overall survival of non-small cell lung cancer, a meta-analysis of published literatures. Lung Cancer 2008;61:91-6. [PubMed]
- Zhan P, Wang J, Lv XJ, et al. Prognostic value of vascular endothelial growth factor expression in patients with lung cancer: a systematic review with meta-analysis. J Thorac Oncol 2009;4:1094-103. [PubMed]
- Wu Y, Liu HB, Ding M, et al. The impact of E-cadherin expression on non-small cell lung cancer survival: a meta-analysis. Mol Biol Rep 2012;39:9621-8. [PubMed]
- Qian Q, Wang Q, Zhan P, et al. The role of matrix metalloproteinase 2 on the survival of patients with non-small cell lung cancer: a systematic review with meta-analysis. Cancer Invest 2010;28:661-9. [PubMed]
- Miao XH, Yao YW, Yuan DM, et al. Prognostic value of the ratio of ground glass opacity on computed tomography in small lung adenocarcinoma: A meta-analysis. J Thorac Dis 2012;4:265-71. [PubMed]
- Mascaux C, Martin B, Paesmans M, et al. Has Cox-2 a prognostic role in non-small-cell lung cancer? A systematic review of the literature with meta-analysis of the survival results. Br J Cancer 2006;95:139-45. [PubMed]
- Begg CB, Berlin JA. Publication bias: A problem in interpreting medical data. J R Stat Soc A 1988;151:419-63.